Laser Marking Systems in Pharmaceutical Serialization

Pharmaceutical serialization is a critical component in safeguarding the global supply chain of medicines. Laser marking systems are proving to be an innovative and reliable solution as counterfeit drugs and compliance regulations continue to challenge the industry.
These systems meet the stringent requirements of pharmaceutical serialization and provide matchless precision, non-invasive application, and long-term efficiency. This post dives deep into the role of laser marking systems in pharmaceutical serialization, its benefits, challenges, and how companies like Tri-Star Technologies are leading the way.
Understanding Pharmaceutical Serialization
Serialization represents the process of assigning a unique identifier to each drug package, enabling traceability throughout the supply chain. This system is vital in combating counterfeit medicines, ensuring patient safety, and meeting regulatory demands from organizations such as the Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Serialization allows companies to track and secure their pharmaceutical products more efficiently than traditional methods, particularly when public trust and regulatory compliance are at stake.
Serialization also provides logistical benefits to pharmaceutical manufacturers and distributors. It improves inventory management, detects diversion within supply chains, and simplifies the recall process. However, it demands precision marking technologies to apply these unique identifiers in a way that maintains the integrity of the medicine.
Laser Marking Systems: An Overview
Laser marking systems are the gold standard for meeting serialization requirements in the pharmaceutical sector. These systems use concentrated laser light to create permanent marks on various surfaces, ensuring durability and readability over time. Unlike other marking technologies, lasers allow for non-contact and high-speed applications tailored for today’s fast-paced pharmaceutical production lines.
How Laser Marking Works
Laser marking focuses a high-energy beam of light onto a substrate to create a photochemical reaction within the material to leave a mark without affecting the structural integrity. Modern systems, such as those developed by Tri-Star Technologies, use advanced software that enables detailed control over the marking process, ensuring precision and compliance with strict regulatory standards.
Types of Laser Marking Systems
Laser systems come in various forms, but UV lasers stand out for their versatility and safety. UV-based solutions are particularly effective for marking pharmaceuticals, as the low thermal energy ensures no damage to the product. Therefore, UV-based laser marking systems are ideal for marking sensitive materials, such as tablets, capsules, and tamper-proof packaging, without compromising the medicine’s integrity.
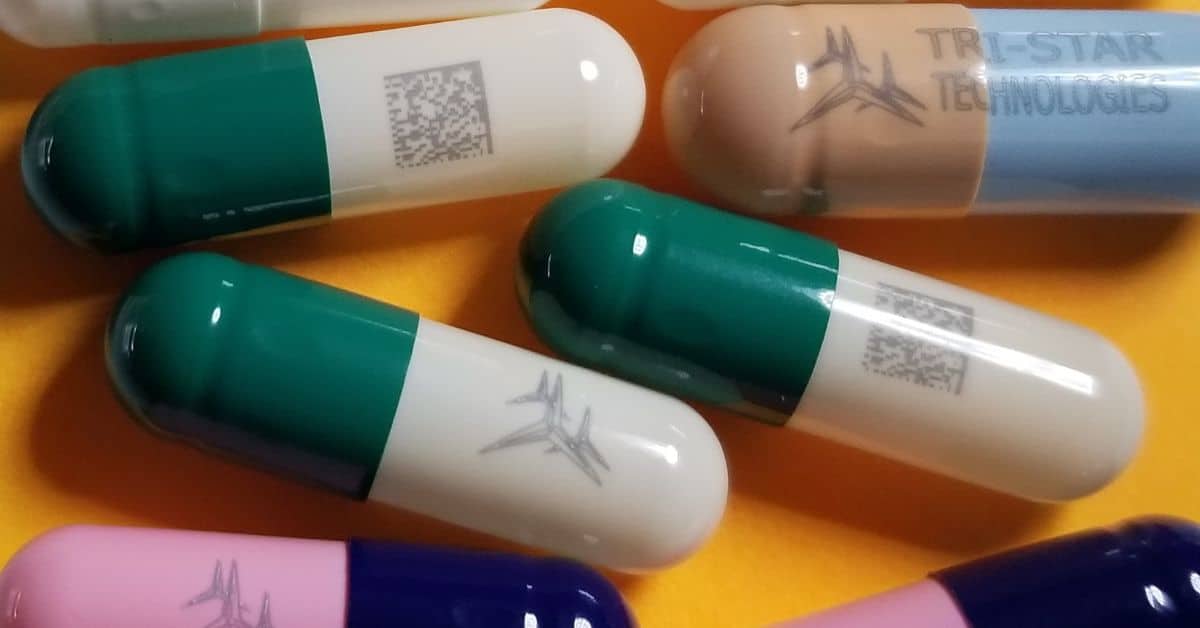
Advantages of Laser Marking
Laser marking outperforms traditional methods, such as inkjet printing or adhesive labeling, in several ways. It’s also more eco-friendly for manufacturers looking for sustainable marking solutions, from producing more durable and precise marks on materials. Consider the following advantages of laser marking:
- Laser marks are permanent, ensuring serial numbers and barcodes remain intact through distribution and handling.
- High-resolution marks maintain clarity even on small or irregularly shaped surfaces, such as pills or caps.
- Unlike ink printing, laser systems are environmentally sustainable as they don’t use consumables.
- UV-based laser marking systems are suitable for sensitive materials, including gelatin capsules.
Laser Marking Applications in Serialization
Effective serialization involves more than simply printing serial numbers on boxes. A good system must address the packaging challenges across the supply chain while maintaining compliance and product integrity. Laser marking meets these expectations in multiple areas.
Marking Tablets and Capsules Directly
Directly marking tablets or capsules with serialization codes aids compliance efforts and deters counterfeiters. Tri-Star Technologies has multiple systems suitable for laser pill marking, such as the M100LPM-R&D and M100LPM150 machines, which can mark tablets’ surfaces precisely without damaging their composition, even for soft gels and other delicate forms.
Marking Through Tamper-Proof Packaging
Many pharmaceutical manufacturers and distributors store their products in tamper-proof packaging, such as blister packs. With cold laser technology, systems like the M100LPM150 can even apply unique identifiers through transparent packaging without compromising its integrity. The ability to mark through tamper-proof packaging ensures that the information remains securely visible without risking damage to the protective seal.
Combating Counterfeiting
Pharmaceutical companies can apply unique, traceable identifiers to individual pills or capsules using an Opioid ID Marker and similar tools. These identifiers enable law enforcement and end customers to verify authenticity when uploaded to a secure database, minimizing misuse and counterfeiting risks.
Tri-Star Technologies Leading the Way
Tri-Star Technologies is an innovator in pharmaceutical laser marking and provides solutions that meet the most demanding industry standards. Tri-Star’s systems are scalable solutions suited for small-scale R&D and full-scale manufacturing and are at the forefront of serialization technology.
Key Products for Serialization
Tri-Star Technologies features multiple systems within its inventory that are ideal for pharmaceutical serialization. Below are examples of laser marking machines suitable for pharmaceutical marking and serialization:
- The M100LPM-R&D is lightweight and portable, perfect for small-scale operations where flexibility is crucial. It offers non-invasive marks on tablets of any size or shape.
- The M100LPM150 is best for large-scale production, as this system delivers high-speed marking capacity while guaranteeing compliance with industry regulations.
- The Opioid ID Marker helps combat the opioid crisis by applying individual identifiers to pills so that authorities can trace the medicine’s origin and distribution.
These systems streamline the serialization process while ensuring drug safety and integrity by combining reliability with cutting-edge features.

Benefits of Laser Marking for Pharmaceutical Serialization
Pharmaceutical serialization using laser marking systems provides unmatched benefits, essential for meeting today’s challenges. Laser marking machines are ideal for pharmaceutical serialization, from providing permanent non-invasive marking to scalability for growing demands.
Permanent Non-Invasive Marking
Unlike ink-based systems that might smudge or peel, laser marking ensures permanence. The process also avoids compromising the structural integrity of delicate products, such as tablets and gel capsules. The non-compromising nature of laser marking means higher-quality marks and less damage to products for a more efficient marking process.
Compliance With Industry Standards
Laser systems, such as those Tri-Star Technologies offer, comply with strict global pharmaceutical regulations. The pharmaceutical industry enforces some of the most stringent compliance standards, so every detail of marking must meet these strict regulations. Companies adopting these solutions can rest assured that they meet requirements for serialization and anti-counterfeiting measures.
Scalability for Growing Demands
Laser marking is available in systems that suit low-volume R&D and high-volume pharmaceutical manufacturing. This scalability is vital for businesses wanting to expand production in response to increased demand or introduce new product lines.
Pharmaceutical Serialization With Tri-Star Technologies
Laser marking systems play a pivotal role in the future of serialization, ensuring the pharmaceutical industry can meet its dual responsibilities of maintaining safety and complying with complex regulations. The possibilities are expanding rapidly with companies like Tri-Star Technologies leading innovation.
Adopting such technology allows businesses to keep pace with regulatory demands and enhance their operational efficiency and reputation for quality. Laser marking is not just the ideal solution for pharmaceutical manufacturers aiming to secure their supply chains, reduce counterfeiting risks, and deliver safer medicines; it is the future. Contact our expert staff at Tri-Star Technologies for more details about our systems and their applications for pharmaceutical serialization.


