The Impact of Laser Marking on Pharmaceutical Traceability
Safeguarding the safety and authenticity of pharmaceutical products is a monumental challenge for the industry. From the manufacturing line to the patient, the journey of a drug involves a complex supply chain vulnerable to counterfeiting, diversion, and errors. These issues pose significant financial risks and, more importantly, threaten patient health. Pharmaceutical traceability, the ability to track and trace medications throughout their lifecycle, is critical to mitigating these dangers.
As regulatory demands for robust traceability systems intensify, manufacturers are turning to advanced technologies to meet them. One of the most promising innovations in this arena is laser marking. This technology offers a precise, permanent, and secure method for identifying pharmaceutical products, providing traceability that traditional methods struggle to match. Keep reading to understand the impact of laser marking on pharmaceutical traceability.
What Is Laser Marking?
Laser marking is a sophisticated process that uses a focused laser beam to create a permanent mark on a material’s surface. This process creates a high-contrast, indelible mark without using any inks, solvents, or other consumables. The interaction is non-invasive and does not compromise the object’s structural integrity.
Applications in Pharmaceutical Traceability
The impact of laser marking on pharmaceutical traceability is significant. Laser marking technology has found several critical applications within the pharmaceutical industry, each contributing to a more secure and transparent supply chain.
Marking Tablets and Capsules Directly
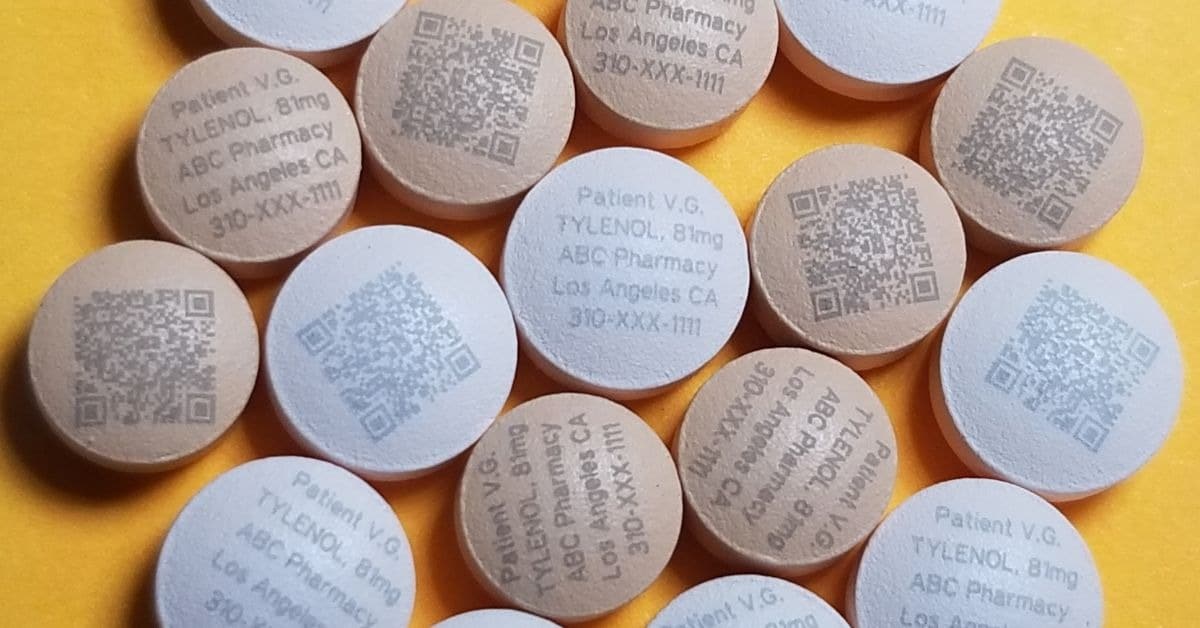
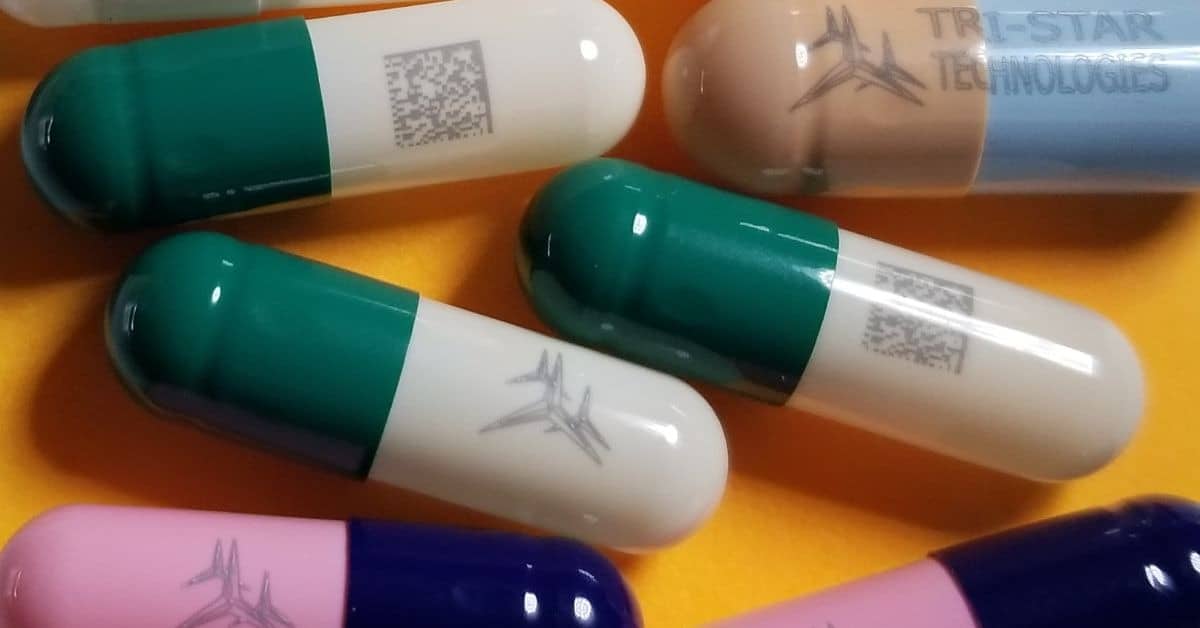
One of the most groundbreaking applications is on-dose marking, where lasers print identifiers directly onto individual tablets and capsules. This capability enables the unique identification of every pill, linking it to its manufacturing batch, prescription details, and intended patient.
Tri-Star Technologies’ systems can mark tablets and gelatin capsules non-invasively, even through tamper-proof packaging material. This on-dose identification is a powerful tool against medication errors and counterfeiting, allowing regulators to identify the identity of a pill at any point, from the pharmacy to the patient’s hand.
Marking on Packaging Materials
In addition to on-dose marking, lasers can also mark primary and secondary packaging materials. This includes applying unique serial numbers, batch codes, and expiration dates onto blister packs, bottles, cartons, and cases.
Laser marks on these materials are high-resolution and permanent, keeping critical tracking information readable for machines and humans throughout the distribution process. This capability is essential for complying with track-and-trace regulations that mandate serialization across the supply chain.
Anti-Counterfeiting Measures
The ability to create unique and permanent identifiers makes laser marking a formidable anti-counterfeiting tool. Manufacturers can use lasers to apply covert and overt marks that are difficult for counterfeiters to replicate.
For example, Tri-Star Technologies’ OPIOID ID Marker is capable of applying any unique identifying information directly onto opioids in either plaintext or QR code. Pharmaceutical companies then can upload this data to a cloud database, allowing law enforcement to trace any pill back to its intended patient, thereby combating misuse and illegal distribution.
Types of Laser Marking Systems for Pharma
Different laser technologies suit various materials and applications within the pharmaceutical industry. The most common types include UV and CO2 lasers. UV lasers, operating at 355nm, are best for many pharmaceutical applications because they enable “cold marking”. This means they minimize thermal stress in the material, which is crucial for heat-sensitive drugs and packaging. CO2 lasers are also common, typically for marking on more robust packaging materials.
Systems by Tri-Star Technologies
Tri-Star Technologies is a leader in developing special laser marking machines for the pharmaceutical sector. Their systems meet the diverse needs of the industry, from research and development to high-volume production.
The M100LPM-R&D is a portable, low-volume system perfect for laboratory settings or small-batch production. Weighing just 32 pounds, this air-cooled laser marker can fit on any tabletop. It can mark tablets and capsules of any shape or size, including softgels and geltabs, without compromising their integrity.
For large-scale pharmaceutical production, the M100LPM150 offers a high-volume solution. This “Cold Laser” system provides the same non-destructive marking capabilities as the R&D model but can also meet the demands of a high-speed manufacturing line. It can even mark pills through the clear window of a sealed blister pack, enhancing efficiency and security.

Advantages of Laser Marking in Pharmaceuticals
The adoption of laser marking offers numerous advantages for pharmaceutical manufacturing and traceability. Laser marking’s permanence and durability enhance pharmaceutical product accountability and patient safety.
Permanent and Non-Destructive Marks
Laser marks are permanent and an integral part of the product itself. They will not smudge, fade, or alter in any way on the material, providing a secure form of identification that lasts the product’s lifetime. Importantly, technologies such as UV laser marking achieve this without generating significant heat, providing a non-destructive mark that does not alter the drug’s chemical composition or efficacy.
Compliance with Regulations
Global regulations, such as the U.S. Drug Supply Chain Security Act (DSCSA), mandate robust track-and-trace systems for prescription drugs. Laser marking enables manufacturers to comply with these regulations by applying unique, serialized codes to each product unit. This allows complete visibility and accountability across the supply chain.
Patient Safety
Ultimately, the most significant advantage of laser marking for pharmaceuticals is patient safety. Clear, permanent identification on both the packaging and the dose itself helps prevent medication errors, guarantees patients receive authentic drugs, and provides a reliable way to verify authenticity. This builds trust and protects patients from the dangers of counterfeit or substandard pharmaceuticals.
Challenges and Considerations
While the benefits are clear, implementing laser marking requires careful consideration. Regulations and material compatibility are key considerations when deciding on a laser marking system for your process.
Regulatory Compliance
Pharmaceutical manufacturing is a highly regulated field. Any new technology must meet Good Manufacturing Practice (GMP) standards so it does not adversely affect the product. Companies must work closely with regulatory bodies to verify that their laser marking processes are fully compliant.
Material Compatibility
Not all materials react to lasers in the same way. It is crucial to test the laser’s compatibility with the specific drug formulation and packaging materials. The goal is to achieve a high-contrast, legible mark without degrading or causing unwanted chemical changes in the product. Companies like Tri-Star Technologies partner with clients to develop custom solutions for their specific material needs.
Advancing Safety with Laser Technology
Laser marking represents a pivotal advancement in the quest for comprehensive pharmaceutical traceability. By providing a means to create permanent, precise, and non-destructive marks on individual doses and packaging, it directly addresses the industry’s most pressing challenges: counterfeiting, medication errors, and regulatory compliance.
As laser systems become more advanced and integrated with digital platforms, their role in safeguarding public health will only grow, solidifying their position as an essential tool in modern pharmaceutical manufacturing. Contact Tri-Star Technologies to explore how our advanced laser marking solutions can integrate into your pharmaceutical manufacturing line, solidifying your commitment to patient safety and product integrity.


