10 Common Reasons Why Medications Are Recalled

Medication recalls are a critical safety measure in the pharmaceutical industry. These actions protect patients from potentially harmful drugs and highlight vulnerabilities in the manufacturing and supply chain. For pharmaceutical engineers and lab managers, understanding the root causes of these recalls is the first step toward prevention.
By reinforcing quality control systems with precision-engineered solutions, companies can safeguard patient well-being and uphold brand integrity. In this article, we’ll explain the most common reasons why medications are recalled and examine how advanced technologies, such as those from Tri-Star Technologies, provide robust solutions to mitigate these risks to safeguard the effectiveness and authenticity of every dose.
1. Manufacturing Errors
Errors during the manufacturing process are a primary driver of medication recalls. Deviations from established protocols can cause incorrect ingredient ratios, resulting in either the active pharmaceutical ingredient (API) being too high, causing toxicity, or too low, rendering the medication ineffective. These errors compromise a drug’s safety, quality, and efficacy, leading to significant financial losses and reputational damage.
2. Labeling Issues
Accurate labeling is a vital component of patient safety. When a medication’s label contains errors, the consequences can be severe. Labeling issues that trigger recalls can range from simple typos to dangerous mistakes, such as an incorrect dosage strength, omitted warnings about side effects, or improper administration instructions. Traditional paper labels are susceptible to damage, misapplication, or removal.
Tri-Star Technologies addresses this challenge with on-dose marking. Our pharmaceutical laser marking machines apply permanent, indelible information directly onto tablets and capsules. This guarantees that the correct identification travels with the medication itself, providing a reliable safeguard against external labeling errors and enhancing patient safety.
3. Contamination
Contamination is one of the most hazardous issues that can necessitate a recall. Pharmaceutical contaminants may be microbial (bacteria, fungi), chemical (cleaning agents), or physical (glass, metal). The source may trace back to raw materials, equipment, or even the marking process itself if it involves inks or solvents. Whichever type of contamination it is or how it gets there, it’s dangerous to consumers and harmful to pharmaceutical suppliers.
Our laser marking systems, including the M100LPM-R&D and the M100LPM150, offer a non-invasive solution that prevents this type of contamination. Because the process uses a cold laser and is entirely contact-free, it introduces no foreign materials to the product. It can even mark a pill through a sealed blister pack, preserving the sterile barrier and protecting the product’s integrity.
4. Stability Problems
Pharmaceutical products must remain stable throughout their shelf life to be effective and safe. Stability problems arise when a drug degrades prematurely, which can reduce its potency or create toxic substances. Factors such as exposure to light, temperature fluctuations, or humidity can accelerate this degradation. Protecting the product from these elements is critical.
By enabling marking through a product’s primary packaging, our laser technology helps maintain the integrity of the seal. This protects the medication from environmental exposure that could compromise its stability, verifying that it meets its specifications until its expiration date.
5. Packaging Defects
Another common reason why medications are recalled is due to packaging defects. A pharmaceutical product’s packaging serves as its primary barrier against environmental factors and contamination.
Defects like broken seals, cracked containers, or the use of improper materials can compromise the medication’s integrity and sterility. A faulty seal on a vial or blister pack could expose the drug to moisture and contaminants, causing degradation. Rigorous quality control of all packaging components is essential.
6. Regulatory Violations
Stringent regulations, such as the Food & Drug Administration (FDA)’s Good Manufacturing Practices (GMP), govern the pharmaceutical industry. Non-compliance with these regulations is a serious offense that can prompt enforcement actions, including mandated recalls. Violations can include failing to adhere to GMP, using unapproved facilities, or making unauthorized changes to a drug’s formulation.
Tri-Star Technologies’ systems support regulatory compliance by providing a reliable method for traceability. Our permanent on-dose marks create a verifiable chain of custody, which is crucial for meeting standards like the Drug Supply Chain Security Act (DSCSA). This helps engineers and quality managers document adherence to protocols and quickly isolate products if an issue arises.
7. Adverse Event Reporting
Post-market surveillance is a cornerstone of drug safety. After the FDA approves a drug, healthcare professionals and patients report unexpected side effects or adverse reactions. If a pattern of severe and previously unknown adverse events emerges, it may indicate a safety issue that requires a re-evaluation of the drug’s risk-benefit profile and, potentially, a market withdrawal.
8. Counterfeit Medications
Counterfeit medications represent a grave and growing threat to public health. These fraudulent products may contain the wrong ingredients, no active ingredients, or incorrect dosages. The global nature of the pharmaceutical supply chain makes it vulnerable to infiltration by counterfeiters, demanding advanced anti-counterfeiting measures.
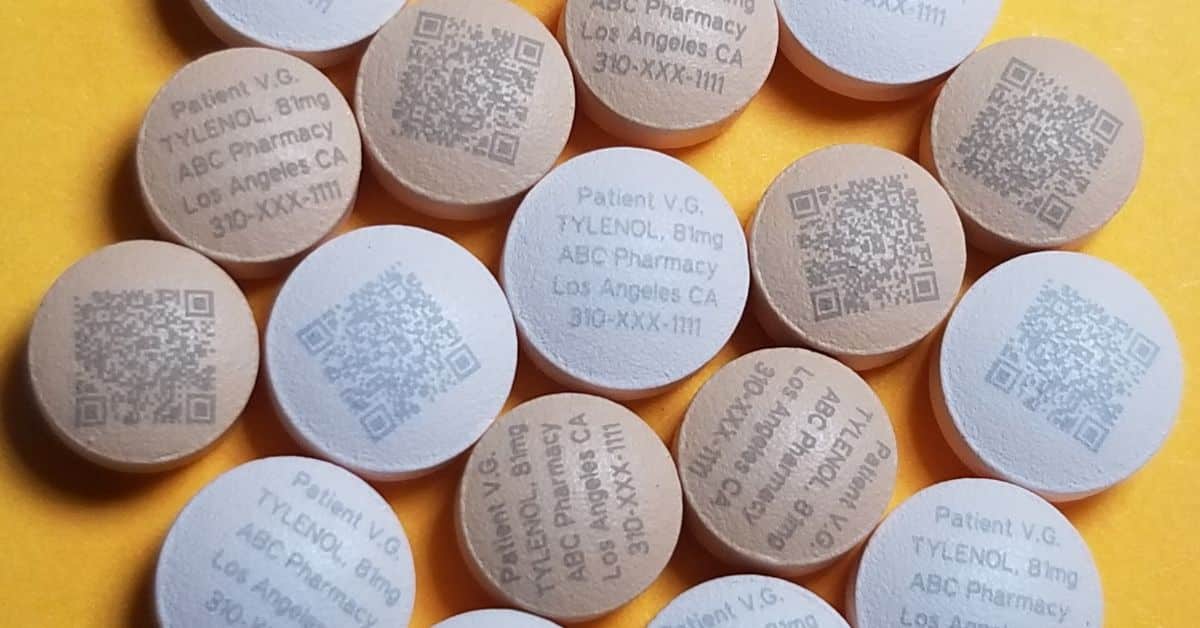
Tri-Star Technologies is at the forefront of combating this threat with our pharmaceutical laser marking equipment. Our patented technology creates permanent, non-invasive marks that are impossible to replicate. A key innovation is our Opioid ID Marker, which applies unique identifying information—such as patient and physician names or prescription numbers—to each dose. Pharmaceutical manufacturers upload this information to a cloud database, allowing law enforcement to trace a pill to its source and verify its authenticity.
9. Supplier Quality Issues
Pharmaceutical manufacturers rely on a network of suppliers for raw materials. If a supplier fails to meet quality standards, it can compromise the final drug product. Impurities in raw materials, variability between batches, or a supplier’s failure to adhere to their own quality control protocols can have a cascading effect that necessitates a recall. Robust supplier qualification and auditing programs are essential.
10. Design Flaws
While less common with oral medications, design flaws are a significant cause of recalls for drug-device combination products like auto-injectors and inhalers. A flaw in the delivery device can prevent the patient from receiving the correct dose or cause injury. These products undergo rigorous testing, but sometimes these flaws make it through quality control processes for the public to discover them, which necessitates a recall to correct the design.
Prioritize Pharmaceutical Product Safety With Tri-Star Technologies
Mitigating the risks that lead to medication recalls requires a multi-layered approach to quality and safety. From confirming label accuracy to deploying advanced anti-counterfeiting measures, every step in a drug’s lifecycle demands unwavering diligence. Tri-Star Technologies offers precision-engineered solutions that empower pharmaceutical companies to enhance product integrity and traceability.
Our advanced laser marking systems provide a permanent, non-invasive, and compliant method for on-dose identification. Contact us today to learn more about our customizable solutions and how they can help you safeguard your products and protect patient health.


